Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ouchi, Kazuki; Komatsu, Atsushi; Takao, Koichiro*; Kitatsuji, Yoshihiro; Watanabe, Masayuki
Chemistry Letters, 50(6), p.1169 - 1172, 2021/06
Times Cited Count:1 Percentile:6.77(Chemistry, Multidisciplinary)The electrochemical behavior of uranium (IV) tetrachloride in ionic liquid-DMF mixture was studied for first time in order to build a redox flow battery (RFB) using U as an electrode active material. We found a quasi-reversible U /U
/U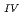 couple that could be applied to the anode reaction of the RFB.
couple that could be applied to the anode reaction of the RFB.
Ouchi, Kazuki; Komatsu, Atsushi; Takao, Koichiro*; Kitatsuji, Yoshihiro; Watanabe, Masayuki
no journal, ,
In this study, the electrochemical behavior of U(IV) in ionic liquid-DMF mixtures was investigated. DMF was added to the ionic liquid to reduce the viscosity to develop the redox flow battery (RFB) based on the reactions of U. The voltammogram of UCl was measured. Redox waves at -1.6 and -1.5 V were observed based on the reaction of U(III)/U(IV). Further oxidation current of U(IV) was not observed at more negative potential than Cl
was measured. Redox waves at -1.6 and -1.5 V were observed based on the reaction of U(III)/U(IV). Further oxidation current of U(IV) was not observed at more negative potential than Cl oxidation. We conclude that U(III)/U(IV) in ionic liquid-DMF mixture is applicable to the cathode reaction of RFB from these results.
oxidation. We conclude that U(III)/U(IV) in ionic liquid-DMF mixture is applicable to the cathode reaction of RFB from these results.
Sugawara, Takanori
no journal, ,
JAEA has started R&Ds on recycling of radioactive waste since 2023. The R&Ds of "Storage Battery Utilization of Unburnable Uranium," "Power Generation by Heat and Radiation," and "Utilization of Elements in Spent Fuels" are being conducted. In this presentation, the background, purpose, technology, current status and future prospects for each item will be introduced.
Ouchi, Kazuki; Komatsu, Atsushi; Takao, Koichiro*; Kitatsuji, Yoshihiro; Watanabe, Masayuki
no journal, ,
The electrode reaction of uranium (IV) tetrachloride in an ionic liquid-DMF mixture was studied in order to build a redox flow battery (RFB) using U as an electrode active material. We found the quasi-reversible U(III)/U(IV) reaction that the formal potential is almost constant at -1.559  0.003 V. This reaction could be applied to the negative electrode reaction of the RFB.
0.003 V. This reaction could be applied to the negative electrode reaction of the RFB.